
The Dr. Nasser Ibrahim Al-Rashid Orbital Vision Research Center is a state-of-the-art laboratory located on the 8th floor of the Evelyn F. and William L. McKnight Vision Research Center at Bascom Palmer Eye Institute. The Al-Rashid Research Center is the first truly translational vision research laboratory of its kind. The Center is exclusively dedicated to the development of novel therapies for some of the most confounding, blinding, and lethal diseases of the visual system.
Directed by David T. Tse, M.D. and Daniel Pelaez, Ph.D., the Center incorporates a diverse array of research scientists, engineers, and clinicians working towards the common goal of translating novel findings from the laboratory bench to the patient bedside. The Center’s underlying goal is to nurture a scientific mindset for multidisciplinary innovation and collaboration by removing barriers and promoting interactions among investigators from different disciplines, supported by the most advanced technologies, facilities, and equipment available.
The Al-Rashid Center is at the cutting edge of biomedical research in ophthalmology with core competencies in the following focus areas:
- Neuroscience and the Optic Nerve
- Orbital Tumors and Malignancies
- Biomedical Engineering
- Stem Cells and Regenerative Ophthalmology
Directorship
David T. Tse, M.D., F.A.C.S.
Executive Director
Dr. Nasser Ibrahim Al-Rashid Chair in Ophthalmic Plastic, Orbital Surgery, and Oncology
Daniel Pelaez, Ph.D.
Scientific Director
Research Associate Professor of Ophthalmology, Biomedical Engineering, and Cell Biology
Center Co-Scientific Director
Dr. Nasser Ibrahim Al-Rashid

Al-Rashid, who lost vision in one eye as a child, first met David Tse 27 years ago and credits him for preserving the sight in his other eye. Through the years they forged a strong connection, which led to Al-Rashid’s generous philanthropy. “Dr. Al-Rashid is a rare blend of intelligence, experience, imagination, and uncommon wisdom,” says Tse. In 2010, Al-Rashid established two endowed chairs at the University of Miami, one to support Tse’s research in ophthalmic plastic, orbital surgery, and ocular oncology at Bascom Palmer; and the other to establish the Dr. Nasser Ibrahim Al-Rashid Chair in the Division of Hepatology and the UM Center for Liver Diseases.
A few years later, the Al-Rashid family made a $10 million gift to build the Dr. Nasser Ibrahim Al-Rashid Orbital Vision Research Center. The grant enabled the construction of the center, and also the recruitment of leading scientists. That support also established two endowments that will ensure scientific research is funded in perpetuity. Dr. Al-Rashid recently committed an additional $2.5 million over the next five years to the Al-Rashid Orbital Vision Research Center.
Neuroscience and the Optic Nerve
A Novel Treatment for Injury to the Optic Nerve
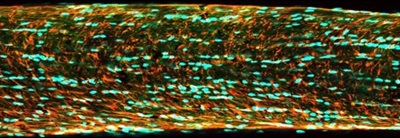
The Al-Rashid Center has recently developed an animal model of TON that closely resembles the human condition without the limitations of tissue ultrastructure damage found in other animal models of optic nerve injury. This model has allowed our scientists to study indirect trauma to the nerve at the molecular level immediately following injury when disease-initiating cascades are first activated. These studies have led to the first-ever targeted treatment for Traumatic Optic Neuropathy, providing patients with the best prognosis for recovering vision. Given the highly encouraging results seen in pre-clinical testing, the Al-Rashid Center is now focusing on the establishment of a large multicenter clinical trial to gain approval of this novel therapy and make it available to all those affected by this devastating condition.
Addressing Mitochondrial Dysfunction in Neurodegenerative Conditions
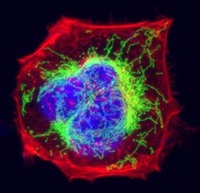
Orbital Tumors and Malignancies
Next-Generation Sequencing of Adenoid Cystic Carcinoma
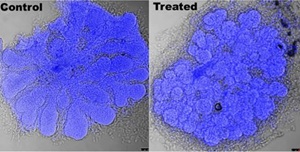
Development of the first Orbital Tumor Oncologic Toolbox
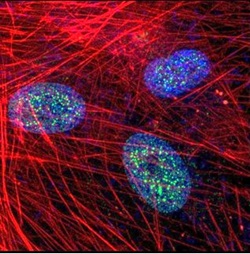
Discovery of Novel Adjuvant Therapeutic Targets in Adenoid Cystic Carcinoma
By studying how tumors respond to the current medical treatments designed to kill them yields powerful insights into the biological mechanisms that cancers use to resist the treatment and stay alive. We have performed studies into how adenoid cystic carcinoma responds to a novel intra-arterial chemotherapy treatment developed by Dr. Tse, and have found novel targets that we can use to augment the effect of the chemotherapy. Using a combinatorial approach, we have shown a strong synergistic effect of this adjuvant treatment in-vitro and in the pre-clinical setting, and are now designing clinical trials to move this new, more effective treatment into the standard of care for these patients.
Biomedical Engineering
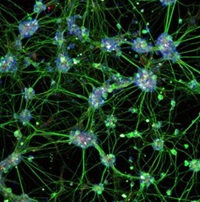
Novel Biomaterials and Compounds for Nerve Regeneration
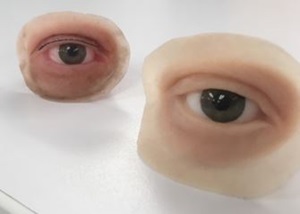 Unfortunately, many conditions in ophthalmology require the removal of the patient’s eye and socket tissues in order to save the patient’s life. For these patients, the resulting facial defect leaves an emotional scar far greater than the physical one, and many cannot afford the cost of a prosthesis. In the Al-Rashid Center, we have devised a way, using state-of-the-art 3D facial scanning and 3D Printing, to manufacture high-quality prosthetics that are both more affordable than the standard prosthetics offered today, and also avoid the timely procedure of mold casting and curing. We have developed this technology also with the hopes of deploying the technology around the world to countries where patients do not even have access to standard prosthetic manufacturers. To further these efforts, we have established a fundraising campaign called Save The Eye (see below).
Unfortunately, many conditions in ophthalmology require the removal of the patient’s eye and socket tissues in order to save the patient’s life. For these patients, the resulting facial defect leaves an emotional scar far greater than the physical one, and many cannot afford the cost of a prosthesis. In the Al-Rashid Center, we have devised a way, using state-of-the-art 3D facial scanning and 3D Printing, to manufacture high-quality prosthetics that are both more affordable than the standard prosthetics offered today, and also avoid the timely procedure of mold casting and curing. We have developed this technology also with the hopes of deploying the technology around the world to countries where patients do not even have access to standard prosthetic manufacturers. To further these efforts, we have established a fundraising campaign called Save The Eye (see below).
Campaign Save The Eye - Bringing hope to patients in need of prosthetic eyes
Save The Eye is a fundraising campaign started by Dr. David Tse and Rick Arkush for the purchase of an ophthalmic 3D printer. Dr. Tse and his team will use this printer to produce financially attainable prosthetic eyes for Bascom Palmer Eye Institute patients, as well as people throughout the world who have lost an eye and cannot afford prosthetics.
There are many people who have lost an eye and then successfully moved on to fulfill their dreams. Rick Arkush is one of them. A patient of Dr. Tse’s, Arkush lost an eye due to a rare form of cancer called lacrimal gland adenoid cystic carcinoma. Rick has now made it a lifelong goal to support positive outcomes for people who have suffered from the loss of an eye or will be faced with that decision.
To contribute to the Save The Eye fund:
- Go to https://advancement.miami.edu/NetCommunity/SSLPage.aspx?pid=28
- Choose “Other” under “Designation”
- Type in “Save The Eye/Account 700523
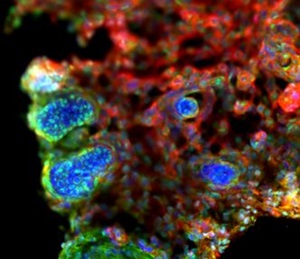
Stem Cells and Regenerative Ophthalmology
The next frontier in human healthcare is Regeneration. At the Al-Rashid Center, we look beyond stopping the progression of disease and have started working towards restoring function and regenerating lost tissues. In no other organ system is the concept of regeneration more impactful to the quality of life than in the eye. That is why we make use of the latest advances in Stem Cell Biology, Genetic Engineering, Epigenetics, Biomaterial Sciences, and Tissue Engineering to drive our core program on Regenerative Ophthalmology. Below are examples of some of our ongoing projects:
Lacrimal Gland Development and Regeneration
Retinal Organoid Platform for Regenerative Cell Therapies
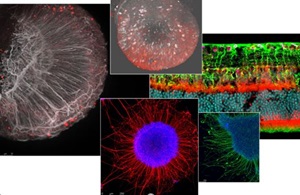
In the eye, cellular replacement therapies are currently under investigation for the treatment of macular degeneration and retinitis pigmentosa. Yet, cellular therapies for neuronal replacement in the inner retina have proven to be more challenging to accomplish. Achievement of a functional regenerative therapy for optic neuropathies would signify a drastic improvement for the quality of life of millions of patients around the world. The Al-Rashid Center team seeks to overcome key technical limitations hindering the development of regenerative therapies for inner retinal neurons in a comprehensive tissue engineering approach to retinal ganglion cell regenerative therapies.
We have developed a stem cell platform that utilizes induced pluripotent stem cells differentiated into retinal organoids in the lab. These organoids help us screen drugs for unforeseen retinal toxicities, but also allow provide us with a source of new retinal neurons to be used in cellular replacement strategies. Our isolated retinal ganglion cells achieve functional maturity and express all of the correct markers for mature RGCs. We are now elaborating on the surgical procedures needed to transplant these new retinal neurons into host retinas and integrate them into the circuitry.

